Leroy Cronin brought media attention to the concept of 3D-printed medications with a 2013 TED Talk discussing the topic.
Since then, the idea has received a great deal of hype, but actual progress has taken place mostly behind the closed doors of research facilities. Only in the past year or so have we seen an increased number of stories revealing that 3D-printed medicine is a bit closer than we might think. This includes not only the first FDA approval of a 3D-printed medication, but also a number of related advances taking place at labs around the world.
Cronin’s lab at the University of Glasgow has published a breakthrough study that may lay the foundations for a new era of 3D-printed medicine. What might that era look like?
Reactionware: A Genie in a 3D-Printed Bottle
Cronin’s concept for 3D-printed medicine is among the first to have reached the ears and eyes of mainstream media. It’s also probably the most complex of the ideas, practically and conceptually, behind 3D-printed drugs.

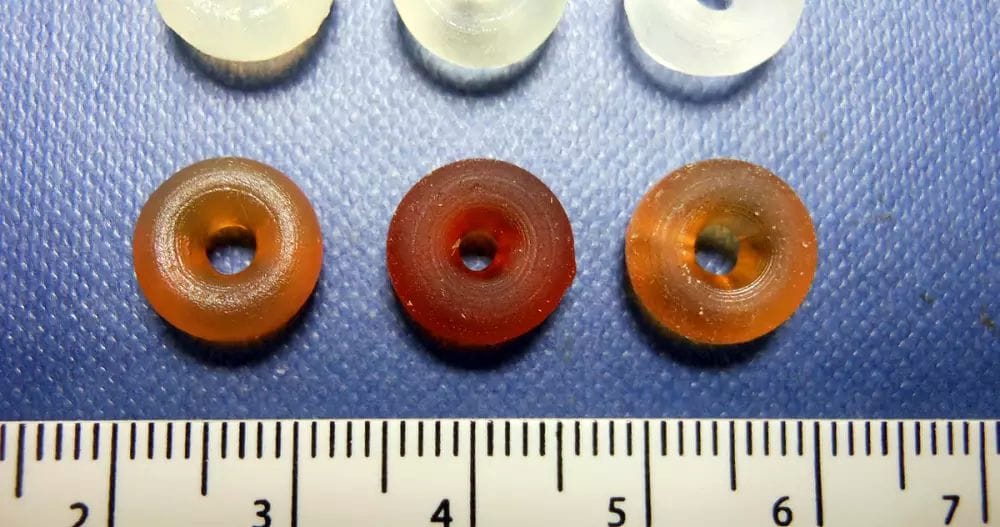
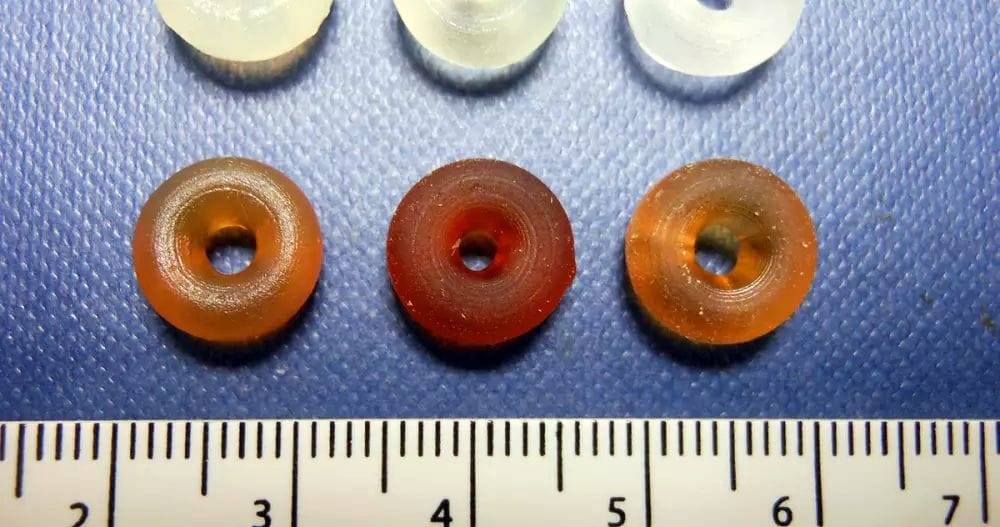
Cronin calls what he and his team make at the University of Glasgow “reactionware,” which more or less recreate the elaborate glassware organic chemists use in researching drug compounds. To create a new chemical compound, Cronin’s lab 3D prints a series of polypropylene containers with openings into which various chemical ingredients, inert gas and air pressure are introduced. When specific chemical agents are inserted into a given module or pushed through to the next using air pressure along a predefined sequence, it’s possible to synthesize a medication.
To demonstrate how the process works, the lab recently developed reactionware for the affordable and accessible muscle relaxant baclofen. The set is made up of five 32 ml modules 3D-printed on an Ultimaker 3D printer. During the printing process, the researchers paused the printer to insert non-printable parts, such as magnetic stirrer bars for reaction modules, fritted glass filters for filtration modules and hydrophobic filters for extraction filters.
Then, over the course of 12 steps—which included the introduction of chemical ingredients into the various modules, as well as heating and cooling various modules and pushing chemistry from one module to the next—the chemists were able to obtain 98 mg of baclofen that was over 95 percent pure, a process that took 40 hours to complete.
Though baclofen is an accessible medication, the use of reactionware could be an important tool for rarer drugs such as photolabels for proteins and tracers used for radiology that degrade quickly and are expensive to produce. Cronin’s lab is working on developing reactionware kits for compounds that can cost around $1 million per mole but could be made much more cheaply with this 3D-printed technology.
Cronin ultimately envisions reactionware as 3D printable by labs and pharmacies on demand. Technicians could download the designs and order a drug-specific kit, made up of prefilled syringes, and produce the medications by following a strict series of steps. The researcher refers to such a process as the “digitization” of medicine, essentially making drug fabrication as easy as following a simple series of steps.
For reactionware to reach this point, Cronin and his team need to ensure that the drug fabrication process is reproducible to exact standards so it can receive approval from the FDA and other regulatory bodies.
Custom Meds from FabRx
Ultimately, one of the goals of 3D-printed pharmaceuticals is custom medication, tailored specifically to the patient. While Cronin’s reactionware may be used for highly specialized drugs, it may be possible to customize more regular medications more immediately.
One startup, FabRx, spun out of and based at University College London, has developed a method for 3D printing custom tablets, or printlets as FabRx refers to them, for producing patient-specific doses. Currently, pharmacies have no way of manufacturing pills in different doses. According to Dr. Alvaro Goyanes, the company’s director of development, by 3D printing medicine directly, it may be easy to modify the dose just by changing the size or infill of the printlet.
“This could be a game-changing technology in pediatric medicine, where the dose of medicines changes depending on the age or weight of the child,” Goyanes said.“On top of that, it will be possible to combine two or more drugs in one tablet, reducing the number of tablets that one person has to swallow. That is important in geriatric populations.”
FabRx is evaluating different technologies for manufacturing medicine, including stereolithography (SLA), selective laser sintering (SLS), inkjetting and fused deposition modeling. While SLA enables extremely high resolution, the resins and photoiniators are not GRAS-approved (generally recognized as safe) by the FDA, according to Goyanes. However, he pointed to research into new resins and polymers that could be biocompatible in the same way that resins used for dental applications are.
As for SLS, Goyanes said, “One of the advantages of SLS is that instead of using plastic, ceramic or metal powder, we can fill the printer with a drug and GRAS-pharmaceutical excipients. The process is very simple, and we get printlets that are perfectly safe. It is not necessary to get a filament by hot melt extrusion like in the fused deposition modeling technology, and, depending on the excipients we select, we can get very fast release or targeted release of the medicines to specific regions of the gastrointestinal tract.”
Read more at ENGINEEING.com