![3D printed electrode design [Source: ScienceDirect]](https://fabbaloo.com/wp-content/uploads/2020/05/image-asset_img_5eb098735d5bb.jpg)
Researchers from Carnegie Mellon and the Missouri University of Science and Technology have developed a method of 3D printing battery components to dramatically increase capacity.
There’s great interest in improving battery technology in recent years due to the ongoing switch from oil to electric power, particularly in vehicle design. Many researchers are investigating a number of improvement avenues, some of which involve tweaking the types of materials involved in battery construction.
But another avenue of investigation deals with the electrodes. In order to increase the capacity of electrochemical reactions, it is desirable to have electrodes with significant surface area. The additional surface area allows the electrolyte more exposure to the chemical process.
In the past, researchers and engineers have used a variety of techniques to achieve increased surface area, such as machining, chemical etching, foaming or other approaches. But this research work involves 3D printing electrodes.
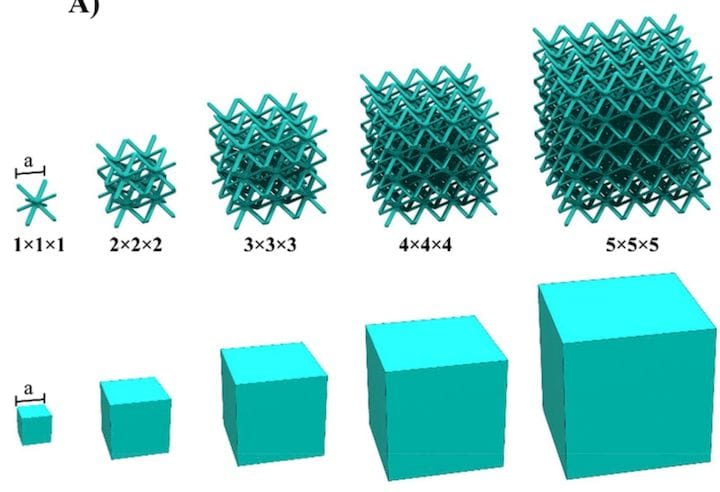
The researchers here devised a three-dimensional structure for the electrode that has significant surface area, and its complex geometry might be familiar to those involved in 3D printing and design. Yes, it’s a lattice structure.
Here we see one of the octohedral structures selected by the researchers for their battery test.
![3D printed deposition techniques for battery electrodes [Source: ScienceDirect]](https://fabbaloo.com/wp-content/uploads/2020/05/image-asset_img_5eb09873b82c3.jpg)
The 3D printing technology used by the researchers was in fact an Optomec 3D printer, one that is capable of 3D printing very small structures. The mixed conductive nanoparticles into the liquid material used by the Optomec device. Once the liquid solidified, the nanoparticles were ready for action.
Conversion to an actual battery was accomplished by producing a sufficiently large set of electrodes and embedding them in a standard CR2032 battery case. Testing of the battery was then performed in a variety of ways.
The researchers produced identically sized electrodes, in block form, as the control for their experiment. These would be similar to what one finds in conventional batteries.
The results showed that the batteries containing the microlattice structures had significantly greater capacity, meaning the number of mAh carried per gram of weight. Results varied depending on the particular structures and sizes of the sub-experiments, but the researchers suggest capacity increases in the range of 400%.
This is a very significant increase. Imagine, for example, an electric vehicle’s range would suddenly increase from 400 km to an incredible 1600 km. Or, as is more likely, the vehicle manufacturers would decide to reduce the number of batteries — and lose a significant amount of weight — and provide a more reasonable range, but with far fewer batteries. In other words, they would be able to make the vehicle far more efficient.
A more interesting development would be for aircraft, which depend utterly on weight. One of the major reasons we have not seen electrically powered aircraft is that the current energy density of battery technology is simply too low to get off the ground; the batteries are too heavy for the energy they provide.
But if batteries had four times the capacity, then it may become technically and economically feasible to develop and offer electrically powered aircraft.
Let’s not get ahead of ourselves, however, as the method of manufacturing these batteries as described by the researchers is obviously very slow and incompatible with mass manufacturing practices. I cannot imagine how expensive the batteries might be if they were literally produced by farms of hundreds of Optomec 3D printers. That’s not going to happen.
However, someone may design a new kind of 3D printer that is specifically engineered to produce this type of 3D structure, and that might be an opportunity for economic feasibility.
Via ScienceDirect








A research thesis details the incredibly complex world of volumetric 3D printing. We review the highlights.