![3D printed microbeams made from living cells [Source: Nature]](https://fabbaloo.com/wp-content/uploads/2020/05/microbeam-fabrication_img_5eb0911d3d0ea.jpg) 3D printed microbeams made from living cells [Source: Nature]
3D printed microbeams made from living cells [Source: Nature]
There’s some very interesting research that could enable breakthroughs in bioprinting by using 3D printed microbeams.
Bioprinting is the science of 3D printing living tissues, and it is the source of the fabled “3D printed organ” stories often found in sensational magazines. The truth is that bioprinting is at a relatively early stage.
3D printing living tissue is a highly complex matter, especially when compared to today’s industrial 3D printing, where typically a single, static material is used to produce an object. By comparison, a bioprint must not only take a specific shape, but must continue to live through absorption of nutrients and release of wastes. Making the matter even more complicated is the fact that living systems grow.
Bioprinting Complexity
Imagine trying to 3D print a highly complex object, made from dozens of materials, that requires an entire interior infrastructure to keep it functioning, and the whole object changes shape over time. That is the dilemma facing bioprinting today.
The researchers explain:
“Mechanical instabilities occur throughout the body at multiple length-scales and stages of life. At large scales, arteries buckle, skin wrinkles, and the growing brain develops deep folds. At smaller scales during development, multicellular epithelial folding and other collective motions coordinate with signaling events, while cell contraction, shape change, and proliferation create stress gradients that produce rugose surfaces and writhing tubes. Controllably facilitating such structural changes in vitro remains a major challenge in engineering tissues and organs. Scaffolds provide predefined structures to guide cell assembly, but the conflicts inherent to simultaneously providing nascent structure and latent plasticity have necessitated scaffolds with increasing complexity in their synthesis, processing, and implementation.“
Microbeams Support Structures
But that job may have just become a bit easier due to some very interesting research from the University of Florida, where work focused on the idea of microbeams.
In a typical bioprint there are two types of materials: the living tissue cells that are the ultimate goal of the print, and a scaffold. The scaffold is a temporary rigid structure on which the living cells can attach themselves to “grow” into the desired geometry.
This is a workable approach, but the idea of using non-living material in a bioprint is not optimal. However, using living cells for structural functions has not been investigated thoroughly until now.
Microbeam Fabrication
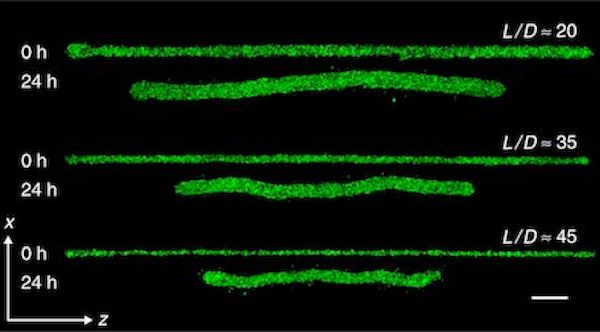
The researchers developed a method for 3D printing microbeams. These are essential “construction beams”, just like you would find at a building site, except that they are fantastically smaller.
The investigation explored how these microbeams would perform, structurally, in a variety of mechanical situations. Specifically, they investigated “how cell-generated forces within these constructs drive deformations, mechanical instabilities, and structural failures”.
In other words, as living cells multiply and grow the overall bioprint, how do the microbeams react to technical stresses generated by other cells? This is a key question that one must answer before being able to design and print more complex biostructures.
Bioprinting Engineering Rules
The research reviewed a wide variety of microbeam structures using different kinds of living materials, some of which were actually cancer cells. They developed what appears to be a rigorous set of methematical rules and formulae that describe the mechanical properties of such structures, similar to what an engineer might use in a life-size building project.
With these new rules, bio-designers have a much better opportunity to develop proper bioprints that can actually grow. Isn’t that the goal of all living things?
Via Nature