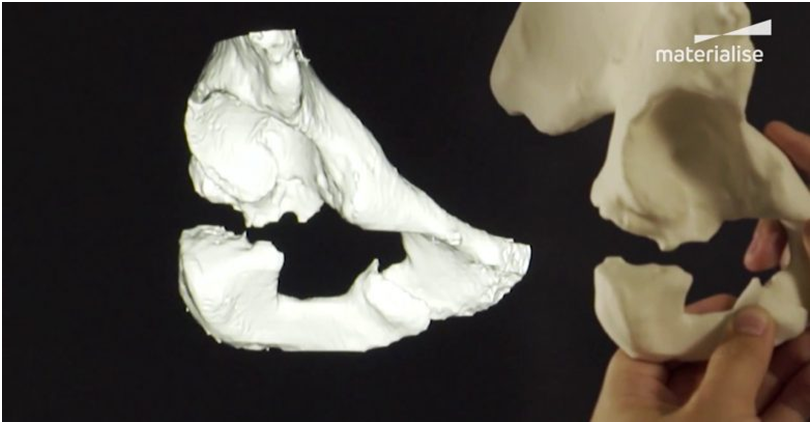
Charles R. Goulding and Mellissa McIntyre consider potential 3D printing implications of Siemens Healthineers’ acquisition of Varian Medical Technologies.
Siemens Healthineers, the 2018 medical spinoff from Siemens, announced on August 1, 2020 the acquisition of Varian Medical Technologies Inc. of Palo Alto, California for an astounding $16.4 billion. The deal will be financed with debt and equity and will close in the first half of 2021.
Both companies have deep X-ray and scanning technology expertise and have already been closely working together in what is called the Envision Partnership. The combination creates an X-ray imaging powerhouse with a holistic treatment approach to cancer and other diseases beginning with planning, all the way to post-operative care. Both companies have 3D printed assets that can be integrated and enhanced.
While the FDA is gradually accepting and approving 3D printed medical products and supplies, Siemens Healthineers and Materialise have already collaborated to bring 3D printing capabilities to hospitals on a global scale. In 2017 they partnered together, to provide the software and resources needed to make anatomical models (using patient data) with added dimensions in the field of medical imaging.
The software developed by Materialise is called Mimics inPrint and it allows radiologists to turn a patient’s body scan data into a digital, 3D printable model. It is compatible with DICOM medical image storage and transmission and can be utilized with all modern medical imaging equipment (e.g., CT and MRI). Siemens Healthineers, with the help of Materialise, has the resources to bring 3D printed medicine to the general population sooner than later.
Varian Medical Systems is a major player in the oncology field having invented the first linear accelerator for targeting tumors with two different types of radiation in 1972. Since then they have developed widely used software systems for treatment planning and imaging devices. A major focus in the medical field is how they can achieve more effective treatments leading to the development of major advances in 3D printing. Additive manufacturing allows medical professionals to provide more customized solutions for medical treatments. In order to make treatments more effective, some researchers focus on personalizing the drug delivery method.
Researchers at the Department of Radiation Oncology, Stony Brook Medicine, Stony Brook, New York studied the efficacy of a bolus produced using a 3D printer. The treatment planning system (TPS) used for the experimental design was Varian Eclipse by Varian Medical Systems. Eclipse is a tool for treatment planning that uses detailed imaging of tumors to develop a method for delivering radiation therapy to a patient. Personalized care has proven to be the main component for improving patient outcomes since everyone may respond to treatments differently due to their physiology. In the experiment, they used the TPS to model the bolus for accurately delivering the treatment using a relatively inexpensive 3D printed solution. This solution is primarily intended for more superficial tumors a treatment delivery is typically hindered by a skin-sparing effect on traditional delivery systems. The skin sparing effect is particularly advantageous for the patient when the targeted tumors are deep, so it avoids damage to the skin. With more superficial tumors, they can overcome this effect by using a bolus, a layer of tissue-like material placed on the patient’s skin. Using a traditional SuperFlab bolus, they found that the treatment is not accurately delivered due to irregularity in the surface of the skin. This irregularity may cause air gaps between the bolus and the surface of this skin leading to some attenuation of the rays. Using Varian Eclipse, they can customize a bolus based on 3D scans of the patient. The customized bolus is then produced using a 3D printer and has been shown to reduce the differences between the calculated doses and the measured doses to less than 1%.
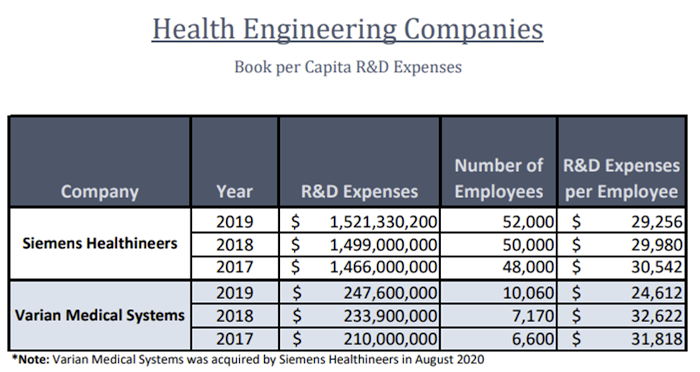
Both companies are research and development (R&D) focused and have similar per capital spends on R&D as well. Time and resources spent on designing and developing 3D printed medical products can be allocated towards certain federal tax benefits.
The Research and Development Tax Credit
Whether it’s used for creating and testing prototypes or for final production, 3D printing is a great indicator that R&D credit-eligible activities are taking place. Companies implementing this technology at any point should consider taking advantage of R&D Tax Credits.
Enacted in 1981, the now permanent Federal Research and Development (R&D) Tax Credit allows a credit that typically ranges from 4%-7% of eligible spending for new and improved products and processes. Qualified research must meet the following four criteria:
- Must be technological in nature
- Must be a component of the taxpayer’s business
- Must represent R&D in the experimental sense and generally includes all such costs related to the development or improvement of a product or process
- Must eliminate uncertainty through a process of experimentation that considers one or more alternatives
Eligible costs include US employee wages, cost of supplies consumed in the R&D process, cost of pre-production testing, US contract research expenses, and certain costs associated with developing a patent.
On December 18, 2015, President Obama signed the PATH Act, making the R&D Tax Credit permanent. Beginning in 2016, the R&D credit has been used to offset Alternative Minimum Tax (AMT) for companies with revenue below $50MM and, startup businesses can obtain up to $250,000 per year in payroll tax cash rebates.
Conclusion
The acquisition of Varian, a world leader in the field of cancer care and research, has created a healthcare colossus. Siemens has justified this merger by pointing out that the prevalence of cancer is expected to double between 2010 and 2030, and over 50 percent of all patients will undergo radiotherapy. Now is the time for 3D printing to take notice of the healthcare industry and imminent changes and acquisitions. Not only will this benefit both the healthcare and 3D printing industries but it can also make a difference in the health outcomes of so many.
Author’s note: Mellissa McIntyre is a graduate of Stony Brook University with a BE in Biomedical Engineering, focusing in Bioimaging and Bioelectricity.
