![Setting parameters on the R1 3D printer [Image: Kumovis]](https://fabbaloo.com/wp-content/uploads/2020/05/kumovis_r1_process_security_img_5eb09220cd068.jpg)
A new medical-focused 3D printer integrates clean room technology.
Medical 3D printing is a fast-growing area. Interest is high, as personalized medical devices like implants can be made ‘at the push of a button.’
Of course it’s not quite that easy, but precise 3D printing is enabling more patient-specific care with on-site technicians capable of creating devices just for an individual. Rather than an off-the-shelf, three-sizes-fit-most model like many medical providers have become used to, 3D printing is enabling a this-size-fits-you model.
Any medical application, though, brings with it more hoops to jump through. As it should: when something is going to be used in contact with or inside the human body, every assurance is needed to ensure the safety of that human.
Kumovis R1



Munich-based Kumovis has now introduced its first production 3D printer with (optional) clean room integration, the R1.
Already on-site with three pilot customers, the R1 is also commercially available as of this month. The system, which looks to be a delta-style extrusion 3D printer, is offered through direct sale or as a subscription model; all availability information (presumably including pricing) is by request.
Key features include homogeneous build chamber heating up to 250°C, with an integrated temperature management system, enabling the use of high-temp materials including the desirable PEEK, as well as generally improved layer adhesion through that temperature control. “Comprehensive monitoring and documentation options” also ensure high process reliability during each print.
The integrated clean room environment is made possible through the temperature control and filtration systems: “In this way, defects caused by foreign particles in the component can be avoided.”
“With the Kumovis R1, we are excited to provide medical technologists with a resource-efficient additive manufacturing system that meets their high requirements, and what is more, leads FLM processing of high-performance plastics to industrial maturity,” said Co-Founder and Managing Partner Stefan Leonhardt. “In addition, with partners such as the software experts from Hyperganic, we will be able to provide rapid access to individualized medical products, and consequently help creating added value for patients and doctors alike.”
Software Partnership
The R1 operates with integrated software from Hyperganic.
Hyperganic’s Managing Director, Lin Kayser, noted that the company “works closely with printer manufacturers” to bring in its software. The partnership with Kumovis brings in Hyperganic Print software-based print control to enhance automation and offer users more print parameter control.
On its About page, the software company notes that:
“Hyperganic builds software to design objects that are as complex, functional, elegant and sustainable as Nature.”
That’s especially helpful — considering the subjects at hand for Kumovis are indeed naturally-designed. Mimicking the functionality and complexity of the human body, even for seemingly simple models and implants, requires exquisite attention to detail, making advanced software a necessity.
Advanced Medical 3D Printing
![A 3D printed cranial implant [Image: Kumovis]](https://fabbaloo.com/wp-content/uploads/2020/05/kumovis_cranial_implant_img_5eb09222415c9.jpg)
Kumovis certainly has its believers already.
In October 2018, the company issued a brief statement:
“We are happy to [announce] that we closed our seed financing round together with High-Tech Gründerfonds and a family office. We will use the 7-digit funding for final product development and the go to market in July 2019.”
Seven digits puts the investment at at least €1 million. While they just barely missed their July goal for market readiness — the R1 was officially available as of 1 August — their strategic approach seems to be essentially on track.
There are a few things this young startup is doing very right: substantial seed funding, strong partnerships, high-temperature capabilities with tight control, application-specific focus, getting equipment into the hands of early adopters.
The Kumovis R1 may just prove intriguing for medical users.
Via Kumovis



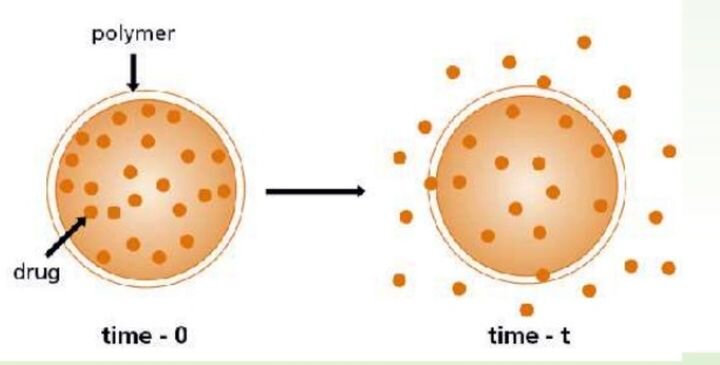

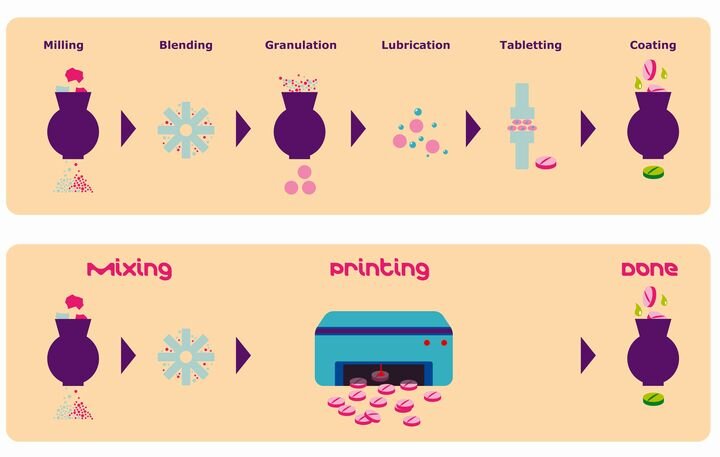




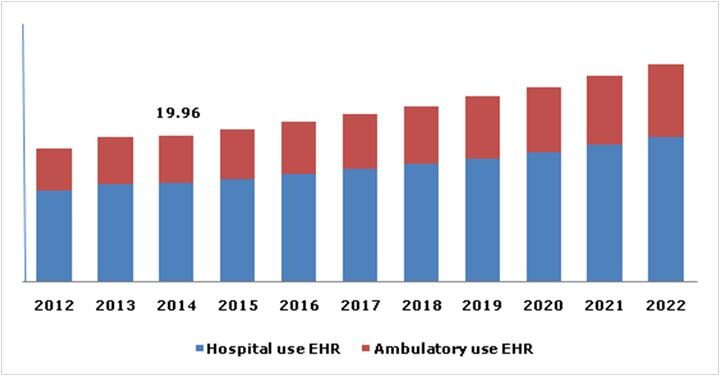
FELIXprinters has released a new bioprinter, the FELIX BIOprinter, which is quite a change for the long-time 3D printer manufacturer.