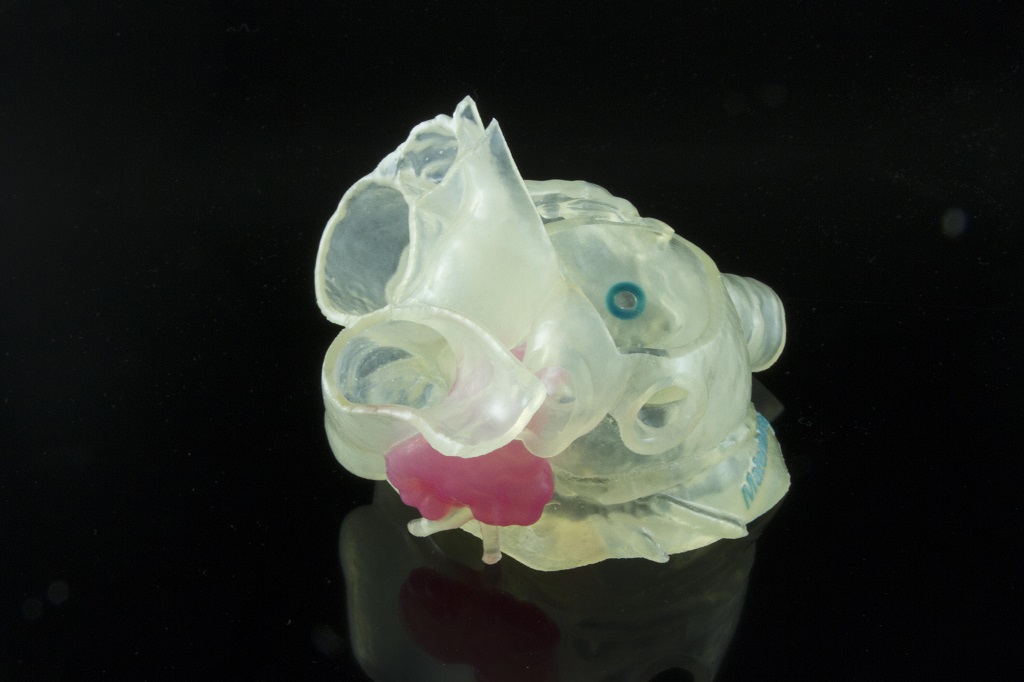
![3D printed model of patient’s left atrial appendage (LAA) created with Stratasys and Materialise technology [Image provided by Stratasys]](https://fabbaloo.com/wp-content/uploads/2020/05/Materialise_cardiac_img_5eb0a1f61a53c.jpg)
3D printing medical models offers benefits to patients and those providing their care with highly accurate patient-specific anatomies able to be held, examined, and practiced on.
Today at RSNA, Stratasys and Materialise are announcing a major relationship focusing on expanding the versatility of materials and machines used in healthcare environments. Stratasys says it is expanding the suite of printers and materials validated by collaborator Materialise with FDA-cleared Mimics inPrint software. [Update: Materialise is also working with Ultimaker and Formlabs through this certiciation program.]
Mimics inPrint received FDA clearance in March 2018, offering end-to-end 3D printing with the all-important FDA 510(k) clearance; to date, it remains the only such software with this clearance for the creation of patient care 3D printed anatomical models. New PolyJet solutions validated through this relationship include the J750 and J735 3D printers as well as the Objet30 Prime desktop 3D printer, as these machines’ multi-color, multi-material capabilities offer new dimensions to 3D printed models.
Of the significance of the expansion of validated offerings, today’s announcement notes:
“The end result is the most versatile 3D printing system for point-of-care across hospitals and physicians – advancing production of patient-specific, life-like anatomical models for diagnostic purposes in conjunction with other tools and expert clinical judgement.”
And:
“According to company reports, of the top 20 U.S. hospitals as ranked by U.S. News & World Report, 16 have implemented a medical 3D printing strategy using Materialise Mimics technology.”
Stratasys is well aware of the value multi-material 3D printing brings to the medical market, while Materialise has long been working toward making 3D printing more accessible directly in hospital environments.
Together, the two companies are looking toward a more integrated medical future — which also fits well with strategies as both Stratasys and Materialise have recently shared with Fabbaloo.
The use of 3D printing in point-of-care facilities is a futuristic offering indeed in healthcare, as creating patient-specific anatomies on-site from scan data generated nearby creates a faster, smoother process bringing together full medical teams working to help a given patient. Patient education is enabled as an individual can hold her own heart in her hands as her physician points to exact areas of concern and can show the exact procedure to be done — and then practice with the medical team to ensure each procedure goes well.
Last year I was on-site at a hospital in Detroit that uses Materialise software to generate cardiac models, and the team driving the initiative had hundreds of stories to share about how these accurate models have helped speed procedure times and reduce failure rates. The benefits of having such models literally in hand cannot be overstated as otherwise internal organs remain digital images on a screen.
“Historically, pre-surgical planning relied on 2D imaging requiring physicians to mentally reconstruct the patient anatomy,” says Eyal Miller, Head of Healthcare Business Unit, Stratasys. “But 3D printing evolves this approach by putting precise replicas of patient anatomy directly in physician hands. Our collaboration with Materialise is a huge step towards unlocking the potential of this technology for patient care. Now the 3D printer that every hospital needs to power their medical modeling comes with additional options for an FDA-cleared software solution.”
Bringing additional colors and materials into play allows for more lifelike, understandable, relatable models to be made. With a science both so exacting and so inexact as human health, every advantage is a benefit; no two hearts are the same any more than any two individuals are the same. Even in well-understood health conditions, symptoms manifest differently and each procedural plan must be tailored to each patient.
“By validating Stratasys’ 3D printing technologies through our certification process, we’re giving doctors and hospitals improved access to high-quality anatomical models for personalized care to patients,” said Bryan Crutchfield, Vice President and General Manager of Materialise North America. “The addition of multi-color and multi-material printers to the list of validated printers is aimed to enable healthcare providers to implement a versatile offering that can support their most complex cases across a wide range of surgical specialties on a single printer. At Materialise, we take a hardware-agnostic approach to software development, offering the flexibility to partner with other leaders in the 3D printing industry like Stratasys – a company committed to addressing requirements of the medical community.”
Companies as large as Stratasys and Materialise combining their expertise to bring realizable solutions to the medical world showcase some of the best of what this technology is capable of, and what this industry at large is capable of. It’s great that motorcycles and rockets can be made better, faster, and lighter — but it’s something else altogether when 3D printing is used to heal and help actual people.
RSNA continues this week in Chicago; Stratasys is appearing at booth 1968L and Materialise in booth 4114, both in the South Hall of McCormick Place.
Via Stratasys and Materialise
[Update 11/26/18: Please find more detail about the Materialise Mimics inPrint Certification program, including a full list of certified hardware and materials, here.]







FELIXprinters has released a new bioprinter, the FELIX BIOprinter, which is quite a change for the long-time 3D printer manufacturer.